Manufacturing and Point of Care Delivery
At Oncofibro our model is building a fast, decentralized manufacturing approach designed to enable median five‑day vein‑to‑vein delivery of reactivated immune cells.
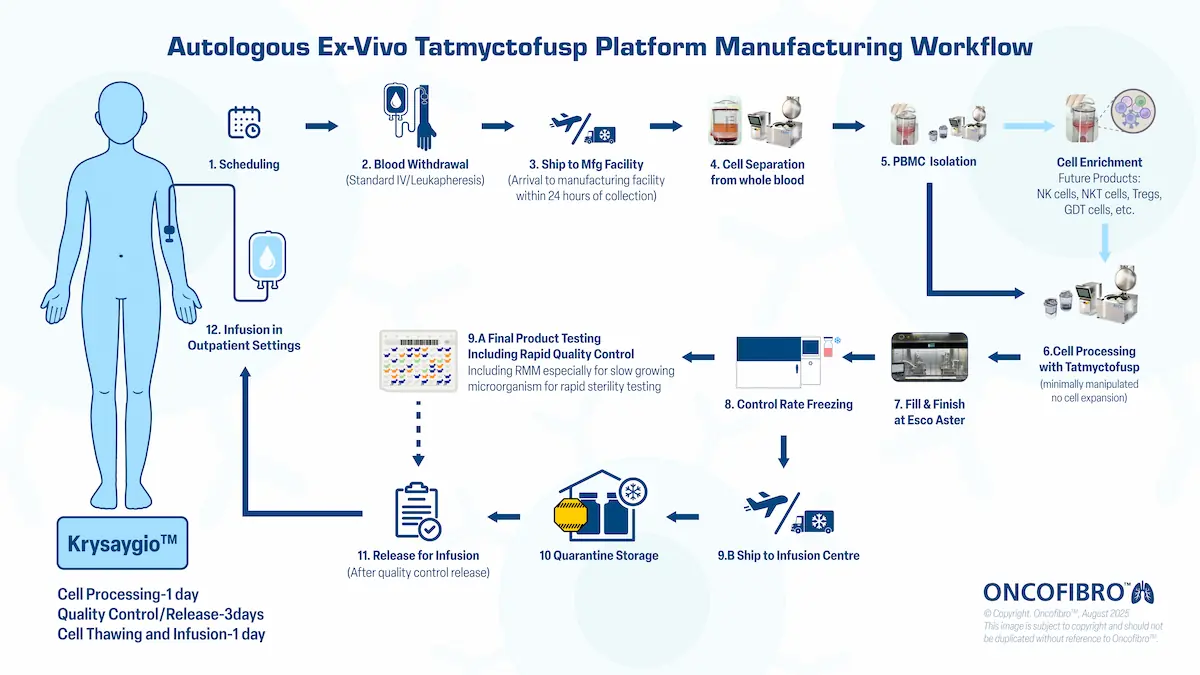
Workflow (targeted timelines)
Day 1 — Cell Processing
PBMC isolation → Tatmyctofusp conditioning (room temperature) →
washing → closed‑vial fill → cryopreservation (same‑day).
Days 2–4 — Quality Control & Release
Rapid sterility and product‑release testing suitable for
point‑of‑care operations.
Day 5 — Thaw & Infusion
Final product prep and administration in an outpatient setting.
Infrastructure & Quality
Closed, single use systems (e.g., “GMP in a box” isolators) to
support decentralized processing. Chain of identity / Chain of
custody with digital batch records and audit trails.
In process controls for viability, phenotype, and potency (assay panel under
development). Release testing: rapid sterility, endotoxin,
mycoplasma (method optimization underway), identity/phenotype,
viability, appearance.
Regulatory alignment: cGMP based procedures, validated cleaning and
environmental controls, and centralized QA oversight for network
consistency.
Scalability & Network Readiness
Standardized SOPs & operator training for multi site deployment.
Technology transfer playbooks to ensure comparability across sites.
Modular capacity planning to match trial demand and future
commercial scale.